Lakes & Climate Change
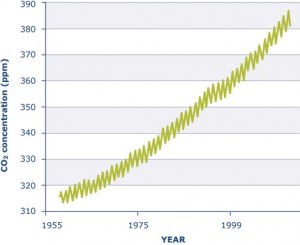
Greenhouse gases, especially CO2, are increasing in the atmosphere due to human activities. Natural sources of greenhouse gases include lakes and other freshwater resources.
Greenhouse Gases
Kevin Rose | Miami University
Gases that trap heat in the atmosphere are known as greenhouse gases. The major greenhouse gases are carbon dioxide (CO2), methane (CH4), and nitrous oxide (N2O)1. At present, atmospheric CO2 is nearly 35% higher than preindustrial levels and is increasing1. Although these gases are released from natural activity, human activity is responsible for the increase of greenhouse gases in the atmosphere. According to both the European Environment Agency and the United States Environmental Protection Agency, CO2 emissions account for the largest share of total greenhouse gas emissions, equivalent to 80-85% of total emissions. Fossil fuel combustion used for transportation and electricity generation are the main source of CO2 emissions, contributing to more than 50% of total emissions.
Natural sources of greenhouse gases include lakes and other freshwaters such as rivers, streams, ponds, and wetlands as well as terrestrial landscapes such as forests and fields. Lakes are active, changing, and important regulators of (CO2), methane (CH4), and nitrous oxide (N2O).
Carbon Dioxide
Carbon Dioxide (CO2) is an important greenhouse gas, and most lakes act as net sources of CO2, releasing it into the atmosphere. Lakes also bury large amounts of carbon in their sediments, over three times more than the world’s oceans.
Lakes play a much greater role in global carbon cycling than their area would otherwise predict. While lakes make up less than 2% of Earth’s surface area, they bury over three times more carbon in their sediments than all of the world’s oceans combined2. This means on a per area basis lakes bury over 100 times more carbon than the oceans. Small lakes that contain large amounts of algae tend to bury the most carbon; thus, the small drainage ponds, farm ponds, and recreational lakes around the world are important sites for carbon cycling and understanding global climate change3.
Despite the fact lakes bury huge amounts of carbon, they also tend to release more carbon dioxide to the atmosphere than they absorb, making them net sources of greenhouse gases. Most of the world’s lakes are supersaturated in CO2 and consequently release some of it to the atmosphere4. In fact, lakes and other freshwaters release almost as much CO2 as all the world’s oceans. This occurs because lakes generally drain large landscapes and the carbon from forests, fields, and lawns becomes concentrated in lakes where it can be buried or released into the atmosphere. Research studies show that the CO2 released from lakes comes from organism respiration — the breathing of bacteria, algae, zooplankton, fish, and other species5.
Methane
While methane is less common than CO2, it is a highly potent greenhouse gas. It has about 20 times more warming potential than CO2. Lakes contribute about 10% of total natural methane emissions, and they produce more methane than the oceans6. Many lakes and other freshwaters produce methane during warm summer conditions or when oxygen levels underwater drop. Most methane is produced in lake sediments when oxygen is no longer present due to different communities of bacteria that grow in environments without oxygen. In some lakes, bubbles can be seen rising from sediments; these bubbles are often methane produced by bacteria in oxygen deprived sediments6. Shallow areas around the shores of warm lakes are hot spots for methane production. Overall, lakes are important sites for carbon dioxide and methane production and release.
Nitrous Oxide
Lakes and other freshwater resources are also sources of nitrous oxide (N2O) cycling, another potent greenhouse gas that is produced in warm lakes by bacteria and other microbes. Within lakes, shallow sediments contribute most to N2O emissions7, while organisms in deep open waters may consume more N2O than they release. Lake shape may be an important predictor of N2O release, as shallow lakes with expansive shorelines may release more N2O than they produce compared with round deep lakes.
Is Hydroelectric Power a “Green” Energy Source?
Lakes and reservoirs are often built or used to generate power. In fact, so much water is retained behind dams that global sea level rise has been reduced by about 0.02 inches (0.55 mm) per year over the past 50 years8. Because fossil fuels are not used to produce hydroelectric power, lakes and reservoirs are often thought of as “green” energy sources. But lakes and reservoirs release potent greenhouse gases — particularly carbon dioxide and methane — into the atmosphere. If a hydroelectric dam releases enormous volumes of greenhouse gases, is it a “green” energy source?

The Three Gorges Dam in China is the world’s largest producer of hydroelectric power. Lakes and reservoirs emit greenhouse gases, and studies have questioned whether hydroelectric is truly a “green” energy source.
Greenhouse gas emissions from freshwater lakes and reservoirs and their contribution to the increase of greenhouse gases in the atmosphere are at the heart of a worldwide debate concerning the electricity generating sector9. Hydropower represents about 20% of the world’s electricity generation capacity and on average emits 35 to 70 times less greenhouse gases per unit power generated than thermal power plants10. When reservoirs are first built for power generation, soils, plants, and trees are flooded. The decay of this plant and soil material can contribute to large emissions of CO2 and CH4 during the first few years after reservoir construction11. Studies show that 3-10 times more greenhouse gases are produced by newly formed reservoirs than from natural lakes of the same size in the first 2-5 years after a reservoir is constructed12.
Beyond the initial release of greenhouse gases, lakes and reservoirs also continue to release carbon dioxide and methane as they produce power. As methane and carbon dioxide enriched water passes through turbines, hydrostatic pressure drops and a large portion of the gas rapidly escapes to the atmosphere. In some regions, such as tropical reservoirs where methane production can be high, reservoirs can release more greenhouse gases than fossil fuel alternatives13.
While lakes and reservoirs used to generate hydroelectric power can release greenhouse gases, they typically release far smaller amounts than traditional fossil fuel based power plants. Hydroelectric is not a perfectly “green” energy source, but it is often much more environmentally friendly than alternative choices and can be part of an alternative energy solutions plan.
Sources:
- IPCC, 2007
- Dean Gorham 1998
- Downing et al. 2008
- Sobek et al. 2005
- Del Giorgio et al. 1999
- Bastviken et al. 2004a
- Huttunen et al. 2003, Wang et al. 2006
- Chao et al. 2008
- Tranvik et al. 2010
- IAEA 1996, Table 1
- Rudd et al. 1993; Kelly et al. 1997
- Tremblay et al. 2005
- Fearnside 1995, 2006










[…] in temperature than are land ecosystems. As organisms respire they produce CO2, an important greenhouse gas. In lakes, increases in temperature increase the rate of CO2 production faster than compared with […]
[…] If a lake is deep enough, water at the bottom will be under so much pressure from the weight of the … […]
[…] carbon dynamics and budget, some have become carbon sinks as opposed to carbon sources. In most healthy systems, lakes ideally function as carbon sources, absorbing large amounts of carbon and releasing more […]