Research Summary: Spatio-Temporal Dynamics Of Parasites Infecting Diporeia Spp. (Amphipoda, Gammaridae) In Southern Lake Michigan, 1980 – 2007
0Amphipods of the genus Diporeia occupy a central position in the food web of the Laurentian Great Lakes ecosystem. As a detritivore that feeds on pelagic material that settles to the benthos, Diporeia are the foremost consumer of primary production in the Great Lakes (Gardner et al., 1990). Because Diporeia are important food resources for numerous Great Lakes fish species, they function as important conduits of nutrients and energy to higher trophic levels and serve as coupling mechanisms between pelagic and benthic zones of the Great Lakes (Fitzgerald and Gardner, 1993).
Historically, Diporeia have been the most widespread and dominant benthic macroinvertebrate in the Great Lakes. Over the last 20 years, however, Diporeia abundances have declined across much of the Great Lakes (Nalepa et al., 1998, 2007; Dermott and Kerec, 1997; Lozano et al., 2001; Barbiero et al., 2011). In the southern basin of Lake Michigan, these declines were associated with increased densities of invasive dreissenid mussels (Nalepa et al., 1998; 2009). Due to the unique position of Diporeia in the food web and the fact that they once accounted for most of the benthic biomass in the lakes, it is believed that these large-scale declines have resulted in major food web restructuring in the Great Lakes (Nalepa et al., 1998).
Although several hypotheses have been proposed, the exact cause of the Diporeia declines remains unknown (Nalepa et al., 2009). Therefore, in an effort to determine the relationship among parasite infection prevalence, invasive dreissenid mussel density, and Diporeia density, we conducted an in-depth quantitative analysis of histopathological lesions found in Diporeia populations from the southern basin of Lake Michigan that had been collected over a period of 30 years (Winters et al., 2014).
Methods
Specimens of Diporeia were randomly subsampled from archived specimens collected between 1980 and 2007 at nine stations in southern Lake Michigan (Fig. 1). For determining infection status and to determine spatial and temporal variability in pathogen presences, Diporeia that had been preserved in 5% buffered formalin were sorted by stage (juvenile < 5.0 mm, adult > 5.0 mm) and processed for histological examination (Fig. 2). Correlation analyses were conducted to assess associations among individual infection prevalences, depth of measurement, Diporeia densities, and dreissenid densities. Correlation analyses were also used to assess associations among several infection prevalences that were identified and depth of measurement, Diporeia densities, and dreissenid densities. To determine if there was any variation in diversity or prevalence of parasitic infections associated with Diporeia populations from before or after the introduction of dreissenids, we analyzed several amphipod samples collected in 1992 and after, representing pre- and post-invasion of dreissenids in the Great Lakes (Nalepa et al., 1998). Models were then fit in an effort to determine the relationship between Diporeia density and infection prevalence as well as to examine other possibly contributing factors, such as dreissenid density.
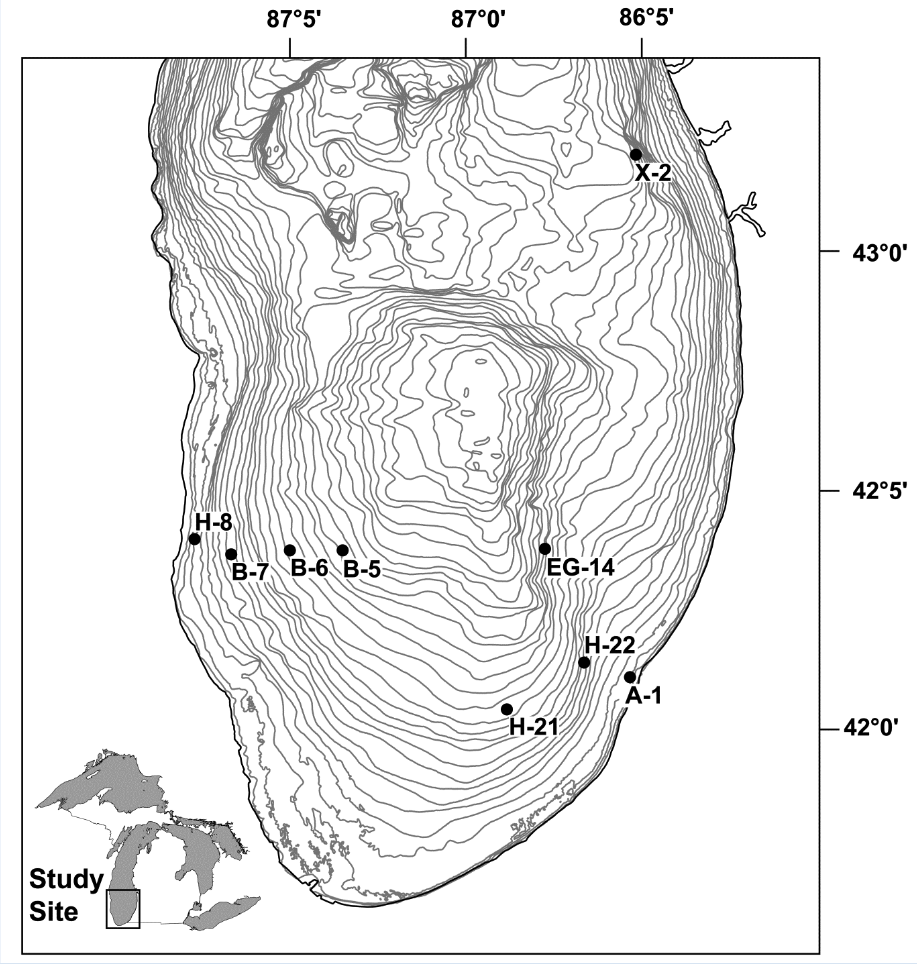
Fig. 1. Location of sampling stations for Diporeia in the southern basin of Lake Michigan from 1980 to 2007. Depth contours are 5 m.
Multiple linear regression was used to relate Diporeia density for each sampling event and location to individual and composite infection prevalences, dreissenid density, and sampling depth. Diporeia and dreissenid mussel densities for each sampling event were obtained from the National Oceanic and Atmospheric Administration Great Lakes Environmental Research Laboratory, Ann Arbor, MI (unpublished data). The base model included dreissenid densities and sampling depth as explanatory variables. We then sequentially added individual and composite infection prevalences (i.e., forward selection) and determined if improvements in model fit justified the increase in model complexity.
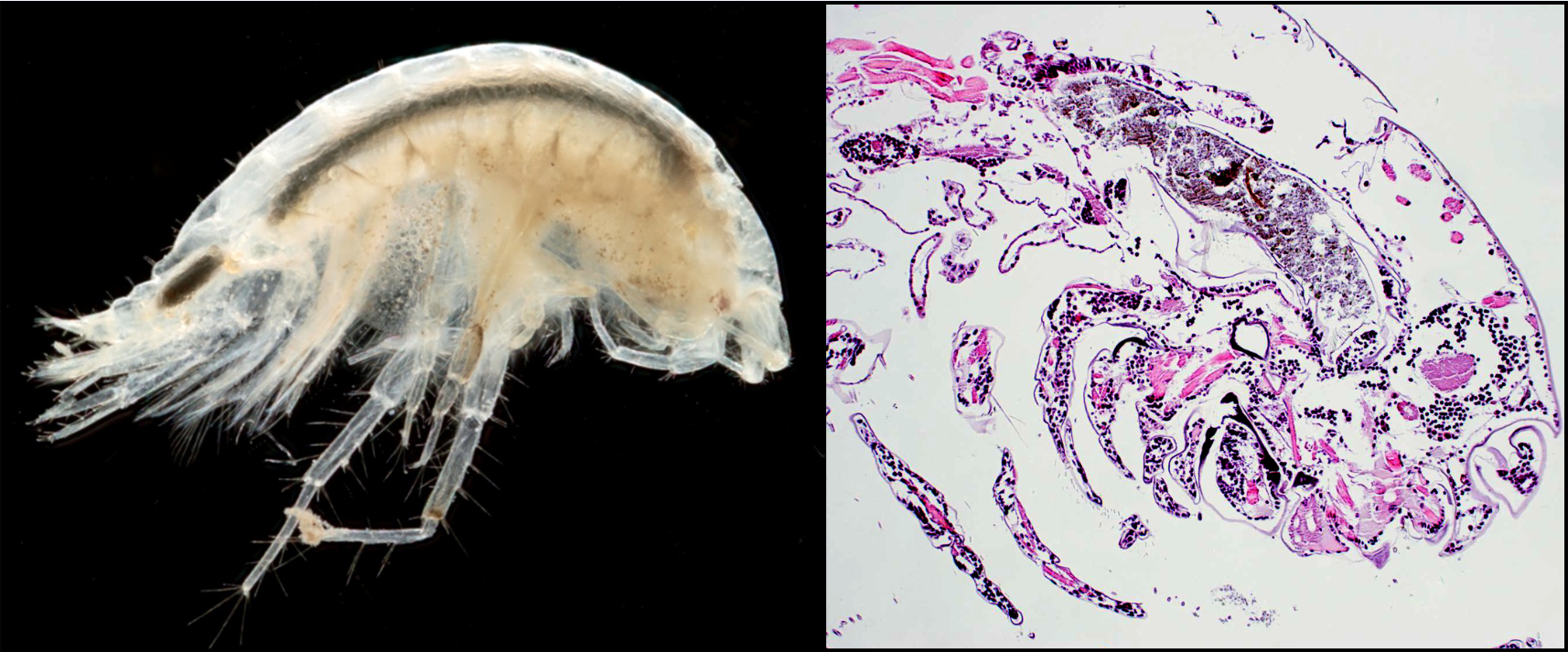
Fig. 2. Diporeia specimen observed under a dissecting scope (left). Stained histological section of a Diporeia specimen.
Results
Several parasites and fungi were identified in the examined Diporeia, including Microsporidia, Haplosporidia, Ciliophora, Gregarinasina, filamentous fungi, yeast, Acanthocephala, and Cestoda. Altogether, 1,624 amphipods (52.2%) exhibited at least one parasitic infection. The reaction to the pathogens varied greatly from differentiated and melanized hemocytic encapsulations in tissues adjacent to parasites to no obvious or negligible responses. A microsporidian and a haplosporidian elicited considerable tissue destruction and host immune response (Fig. 3).

Fig. 3. Histological section of a haplosporidian (left) and a microsporidian (right) in infected Diporeia sp.
Spatio-temporal variability in parasitic infections was observed with prevalences often fluctuating by depth, sampling site, and life stage of Diporeia (Fig. 4-5). Statistically significant positive correlations were observed between prevalences of acanthocephalan infections and microsporidian infections (t=0.355, P=0.002), prevalences of haplosporidian infection and yeast infections (t=0.365, P=0.004), prevalence of acanthocephalan infection and sampling depth (t=0.208, P=0.049), prevalence of haplosporidian infection and Diporeia density (t=0.226, P=0.028), and prevalence of filamentous fungi infection and dreissenid density (t=0.244, P=0.035). Conversely, statistically significant negative correlations were observed between prevalence of Gregarinasina infections and dreissenid density (t=-0.278, P=0.007), Diporeia density and dreissenid density (t=-0.478, P<0.001), Diporeia density and sample depth (t=-0.227, P=0.012), and dreissenid density and sample depth (t=-0.211, P=0.036). Statistically significant negative correlations were observed between prevalence of co-infections and dreissenid density (τ = -0.291, P=0.004). Increased infection prevalences for multiple parasitic infections prevalences were observed in samples collected in 1992 and 1993. The best fitting multiple linear regression model included dreissenid density, sampling site depth, and microsporidian infection prevalence.
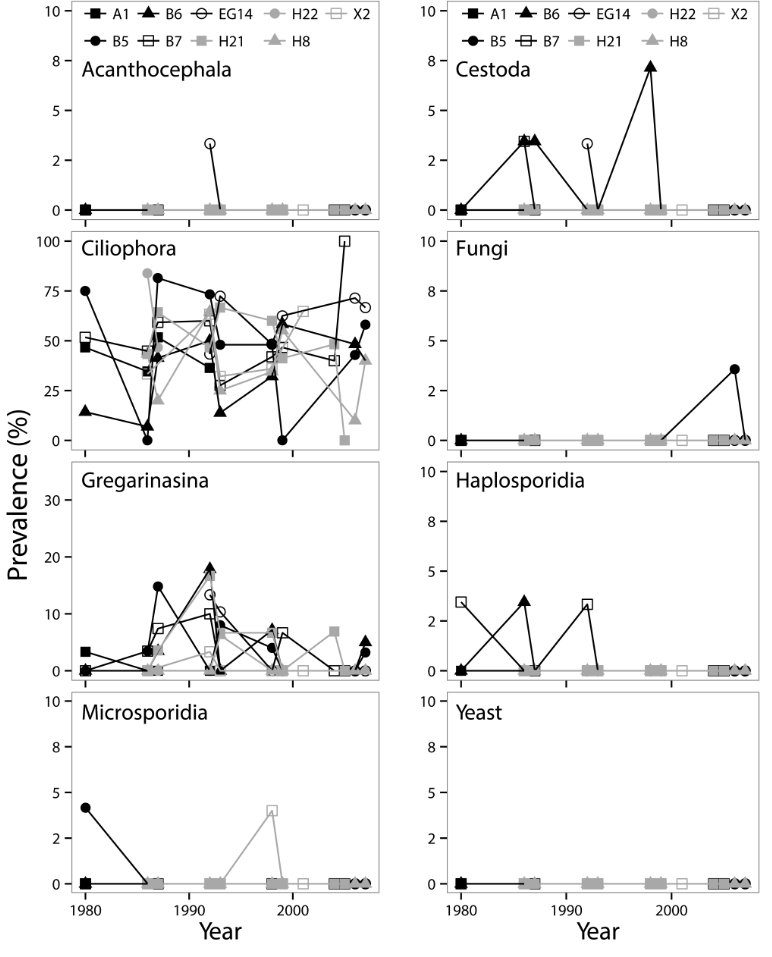
Fig. 4. Prevalence of parasitic infections in juvenile Diporeia spp. collected from nine sampling sites in the southern basin of Lake Michigan between 1980 and 2007.
Conclusions
Through this work, we were able to determine a number of distinct relationships among Diporeia density, parasite transmission efficiency, age of Diporeia, dreissenid mussel density, and temporal and environmental factors. Additionally, we were able to select a model that describes trends in Diporeia density in the southern basin of Lake Michigan over the past three decades in relation to physical and biological factors. Model testing showed a strong correlation between dreissenids density and Diporeia density, however, no distinct increase in a particular parasite infection(s) could be linked to increased dreissenid density or decreased Diporeia density. Interestingly, an increase in infection prevalences was associated with the establishment of dreissenids in Lake Michigan suggesting there is evidence to suggest a mechanistic link exists between dreissenids and the decline of Diporeia. The finding that prevalences of co-infections by multiple parasites showed a significant negative correlation with Diporeia density suggests it is plausible that the interrelationship and dynamics of different parasitic infections may synergistically contribute to the decline in Diporeia populations in the southern basin of Lake Michigan.
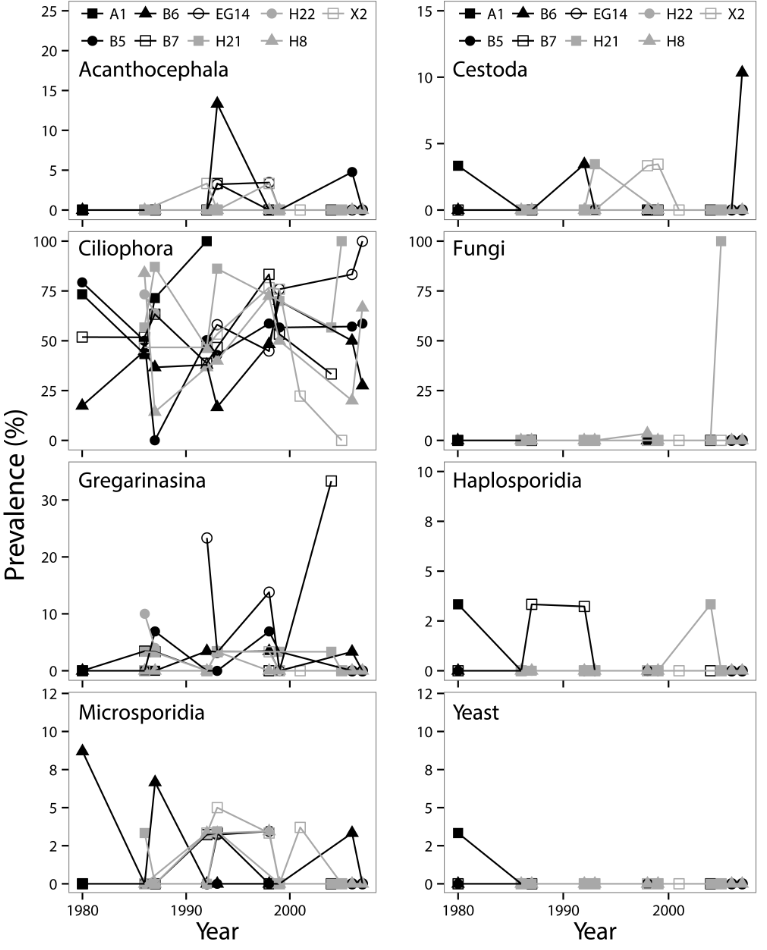
Fig. 5. Prevalence of parasitic infections in adult Diporeia spp. collected from nine sampling sites in the southern basin of Lake Michigan between 1980 and 2007.
In two separate studies, we performed further ultra-structural and molecular studies on the observed haplosporidian and microsporidian (Winters and Faisal, 2014a; 2014b). These studies clearly revealed that both parasites are novel. The haplosporidian which caused systemic infection bears some similarities with the oyster pathogen Haplosporidium nelsoni. The microsporidian which caused severe muscle destruction and likely impairs the normal movement, feeding, swimming, and overall functioning and fitness of Diporeia bears some similarities to the genus Dictyocoela, a group of microsporidians that are known to infect amphipods.
Collectively, the findings of these studies provide valuable insights not only on the identity and dynamics of parasites infecting Diporeia during the declines but also on the potential transmission of parasites that use Diporeia as an intermediate host. This new knowledge is needed to understand the potential causes of the decline in Diporeia and foster the development of efficacious management strategies for the restoration and conservation of Diporeia and other ecologically important organisms in the Great Lakes food web.
Full study published in the Journal of Invertebrate Pathology, September 2014, Vol. 121, p. 37-45
Selected References
- Barbiero RP, Schmude K, Lesht BM, Riseng CM, Warren GJ, Tuchman ML. 2011. Trends in Diporeia populations across the Laurentian Great Lakes, 1997-2009. Journal of Great Lakes Research. 37: 9-17.
- Dermott R, Kerec D. 1997. Changes to the deepwater benthos of eastern Lake Erie since the invasion of Dreissena: 1979-1993. Canadian Journal of Fisheries and Aquatic Sciences. 54: 922-930.
- Fitzgerald SA, Gardner WS. 1993. An algal carbon budget for pelagic-benthic coupling in Lake Michigan. Limnology and Oceanography. 38: 547-560.
- Gardner WS, Quigley MA, Fahnenstiel GL, Scavia D, Frez WA. 1990. Pontoporeia hoyi-a direct trophic link between spring diatoms and fish in Lake Michigan. In Large lakes Springer Berlin Heidelberg pp. 632-644.
- Lozano SJ, Scharold JV, Nalepa TF 2001. Recent declines in benthic macroinvertebrate densities in Lake Ontario. Canadian Journal of Fisheries and Aquatic Sciences. 58(3), 518-529.
- Nalepa TF, Hartson DJ, Fanslow DL, Lang GA, Lozano SJ. 1998. Declines in benthic macroinvertebrate populations in southern Lake Michigan, 1980-1993. Canadian Journal of Fisheries and Aquatic Sciences. 55: 2402-2413.
- Nalepa TF, Fanslow DL, Lang GA. 2009. Transformation of the offshore benthic community in Lake Michigan: recent shift from the native amphipod Diporeia spp. to the invasive mussel Dreissena rostriformis bugensis. Freshwater Biology. 54: 466-479.
- Nalepa, TF, Fanslow DL, Pothoven SA, Foley AJ, Lang, GA. 2007. Long-term trends in benthic macroinvertebrate populations in Lake Huron over the past four decades. Journal of Great Lakes Research. 33: 421-436.
- Winters, A. D., Fitzgerald, S., Brenden, T. O., Nalepa, T., Faisal, M. 2014. Spatio-temporal dynamics of parasites infecting Diporeia spp. (Amphipoda, Gammaridae) in southern Lake Michigan (USA). Journal of Invertebrate Pathology, 121: 37-45.
- Winters, AD, Faisal, M. 2014a. Molecular and ultrastructural characterization of Haplosporidium Diporeiae n. sp., a parasite of Diporeia sp. (Amphipoda, Gammaridea) in the Laurentian Great Lakes (USA). Parasites & Vectors, 7: 343.
- Winters AD, Faisal M. 2014b. Molecular and ultrastructural characterization of Dictyocoela Diporeiae n. sp.(Microsporidia), a parasite of Diporeia spp.(Amphipoda, Gammaridea). Parasite, 21, 26.
Featured image: View of Lake Michigan and downtown Chicago as seen from Fullerton Drive and Lake Shore Drive. (Credit: Flickr User Ron Cogswell via Creative Commons 2.0)













